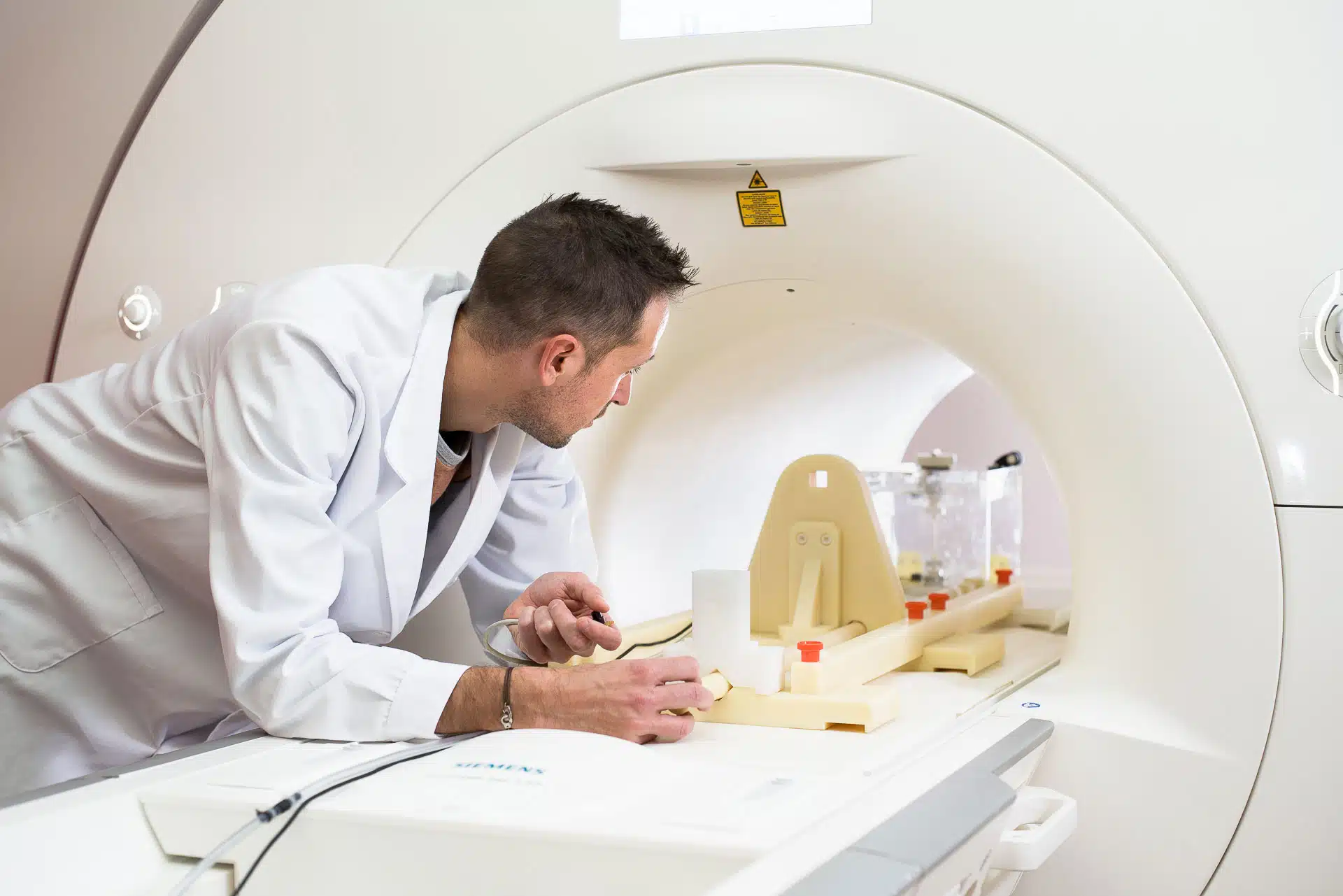
Have you already conducted a comprehensive assessment of the MRI safety and MRI compatibility of your medical devices?
Evaluating MRI safety is a crucial step in the entire lifecycle of a medical device. It not only influences clinical use but is also a key requirement for regulatory approval and successful market access in Europe, North America, and worldwide.
As a leading expert in MRI safety and compatibility, Healtis combines scientific excellence with deep regulatory insight. Our team actively contributes to the development of MRI safety standards and advances in the field, ensuring we remain at the forefront of knowledge regarding interactions between MRI systems and medical devices.
Through close collaboration with manufacturers worldwide, we have also built strong expertise in international regulatory expectations. This unique combination makes Healtis an ideal partner to help you achieve a robust, compliant, and well‑justified MRI safety evaluation.
Our support can include accredited testing, numerical modeling, technical and regulatory guidance, and assistance with device design and labeling.
If you need support in evaluating the MRI safety of your devices, we are here to help.
Feel free to contact us:
Spencer PARENT – [email protected] (North American time zone)
Thibault KLAMMERS – [email protected] (European time zone)
We help you ensure the safety of your products and accelerate the path to successful market launch.