HomePage
Recent Postss
Read more

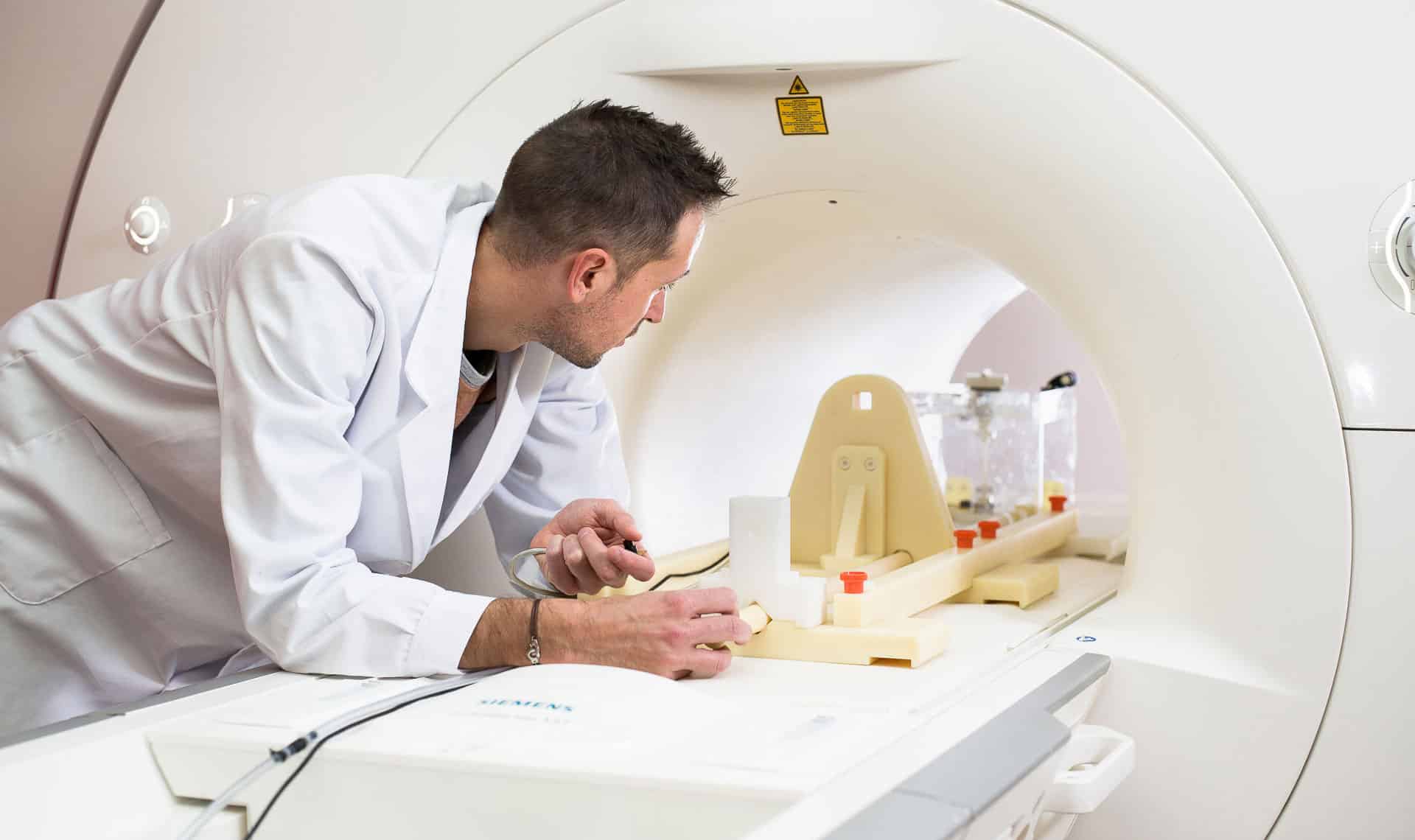
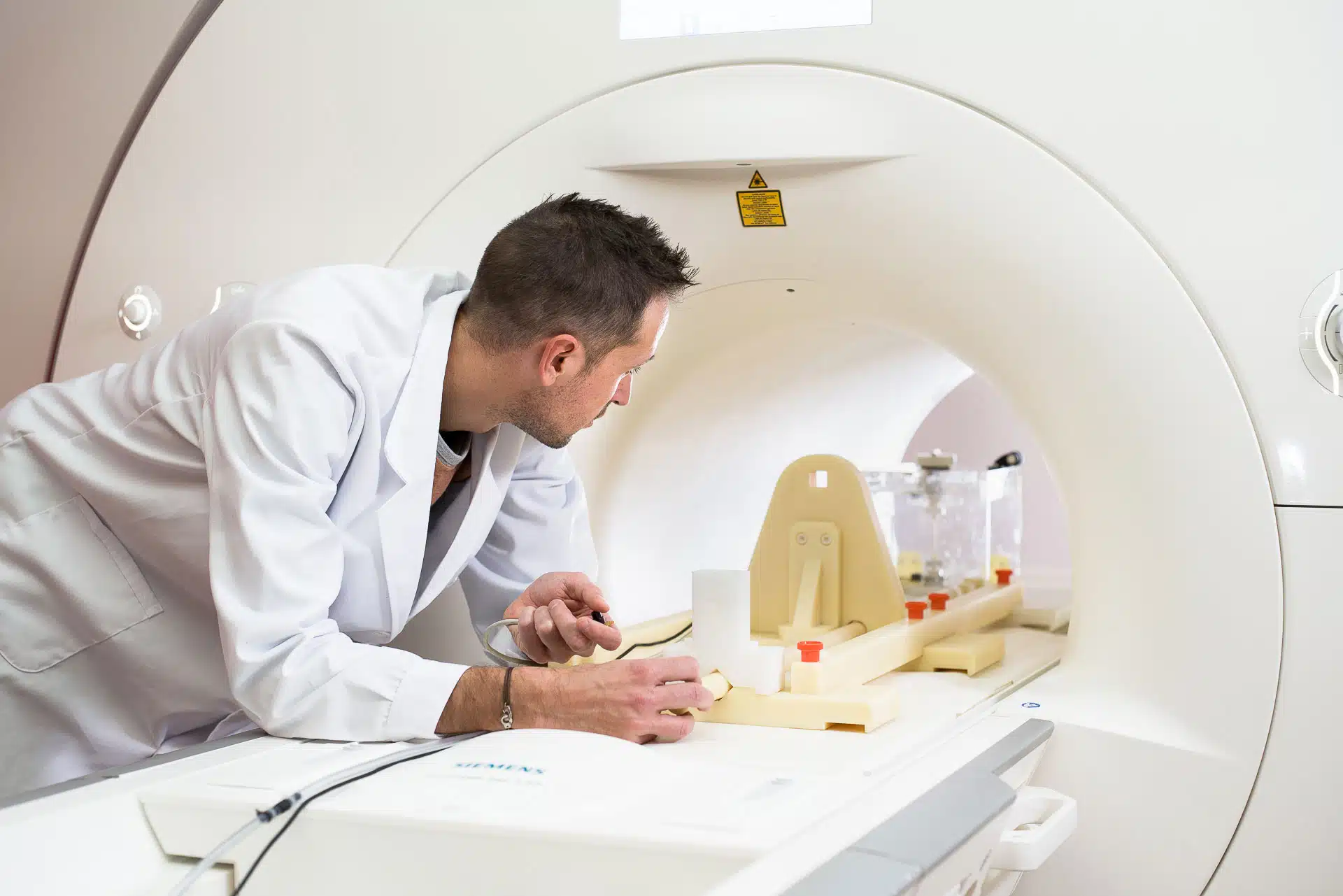
MRI safety and compatibility testing
We offer a wide range of tests for evaluating the safety of medical devices in the MRI environment +
Read more

Numerical modeling
Numerical modeling allows the determination of a worstcase in a multi-feature device array. +
Read more
R&D Support
MRI involves complex and demanding physical phenomena. Designing a device compatible with MRI can be long and expensive. +
Read more

Technical and regulatory expertise
Risk assessment in MRI and development of a strategy for managing these risks. Marking aid (ASTM F2503) (...) +
Read more

Training services
Our trainings, delivered by our experts, are intended to anyone who may enter the MRI environment. +
Read more

Our commitments
HEALTIS is accredited by COFRAC, according to standard NF EN ISO / CEI 17025 for its testing activities +
Newsfeed
Read more
Numerical modeling: reliability, experience and dedicated tools
(article updated on May, 5th 2026) What is the role of numerical simulation for medical device heating in MRI environments? Introduction In an MRI environment, radiofrequency (RF) electromagnetic fields can induce sig... +
Read more
Gradient-Induced Vibrations : HEALTIS Published in MRM!
HEALTIS Work Published in Magnetic Resonance in Medicine! HEALTIS is delighted to share that our work on experimental measurements of gradient-induced vibrations in MRI has been published in *Magnetic Resonance in Med... +
Read more
MRI-induced vibration on your medical devices: assessment methods & expert opinion
New MRI vibration assessment method now available at Healtis Healtis enriches its test catalog by integrating the Tier 2 method for evaluating vibrations induced by MRI gradients, in accordance with ISO/TS 10974:2018 ... +
Read more
Healtis will be present at AAOS 2026 in New Orleans!
Our representative Spencer Parent will be onsite in New Orleans this week to meet with Orthopedic manufactures to discuss the importance of MRI Safety and Compatibility testing! Have an implantable medical device for ... +