Healtis Featured in DeviceMed: Spotlight on Our MRI Risk Assessment Strategy Service
DeviceMed has published an article highlighting one of our key services: expert support for defining an effective MRI risk assessment strategy for medical devices.
With Medica / Compamed trade-fair approaching, this feature underscores the growing importance of MRI safety in the development and regulatory approval of medical technologies.
What DeviceMed Highlights About Healtis
The article begins by recalling the origins of Healtis, founded in 2012 and rooted in the IADI Inserm U947 and CIC‑IT Inserm CIT801 research laboratories. These teams bring over 20 years of experience in designing instruments for the MRI environment. DeviceMed emphasizes the strong expertise and recognition Healtis has built over the years, driven by our commitment to service quality, customer support, and continuous R&D.
It also outlines the range of services we provide, including:
-
support for designing MRI‑compatible medical devices,
-
numerical modeling to predict risks or identify worst‑case scenarios,
-
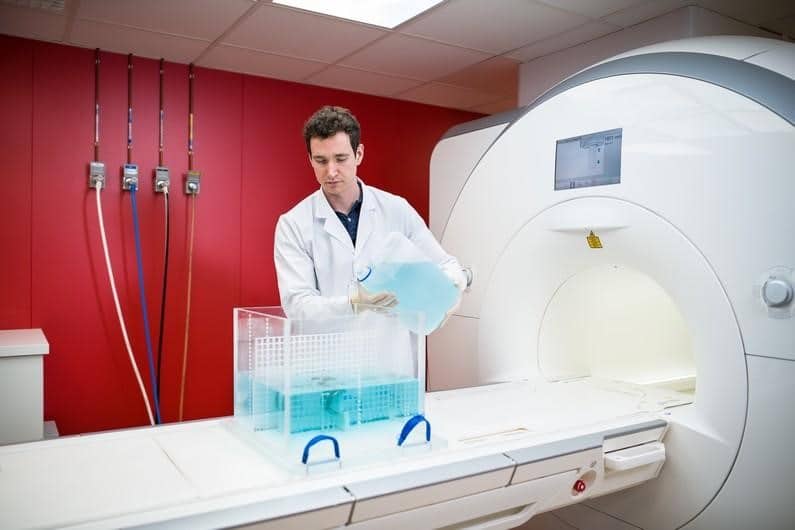
MRI safety and compatibility testing under ISO 17025 accreditation,
-
interpretation of test results and assistance with ASTM F2503 labeling.
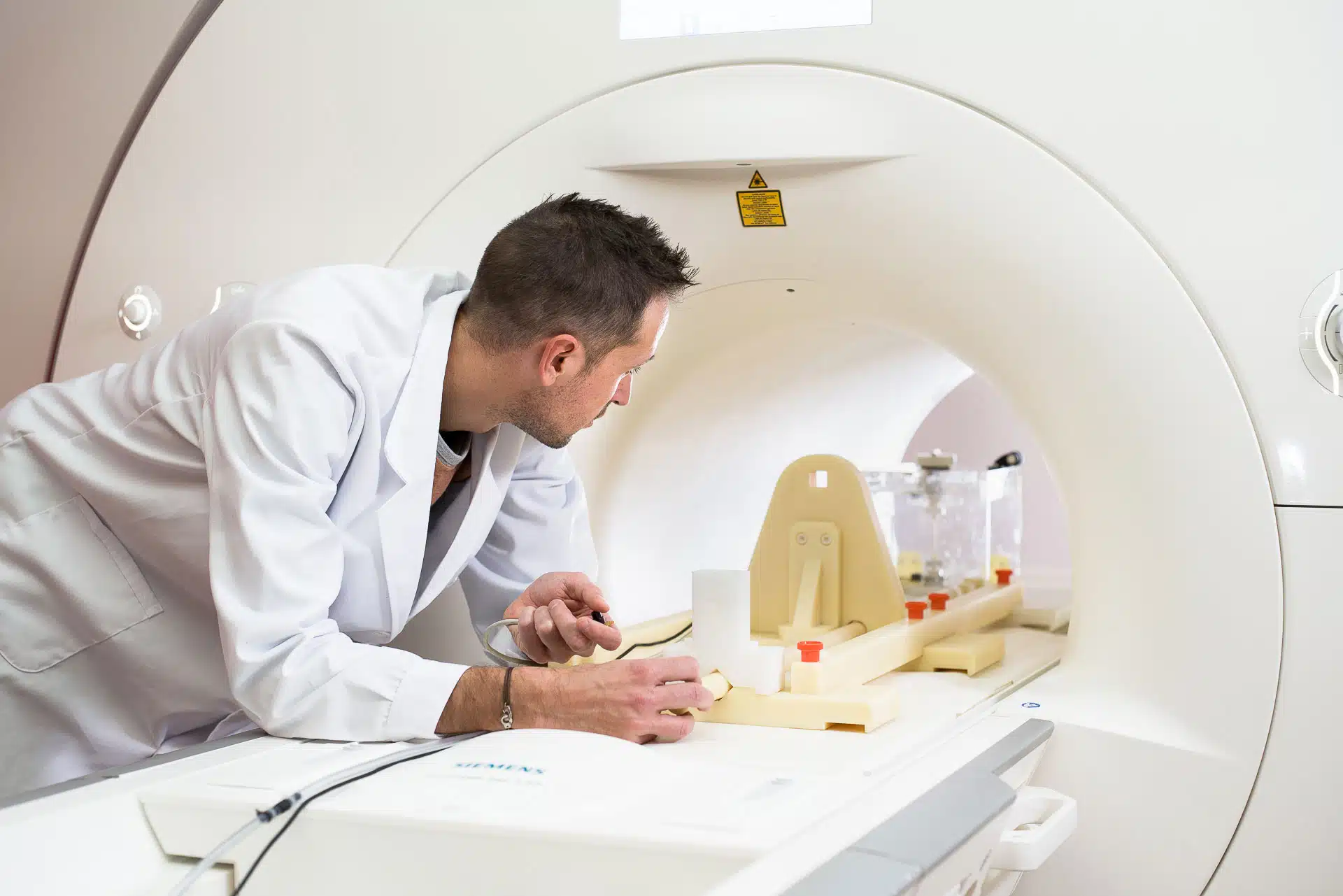
The article focuses particularly on our “MRI safety assessment strategy definition” service : a crucial first step in conducting a compliant and device‑appropriate MRI risk study.
What the Strategy Definition Service Includes
DeviceMed describes the service as a structured, expert‑guided process designed to help manufacturers navigate the complexities of MRI safety. The service includes:
-
a detailed review of the device (materials, composition, configurations, MRI use conditions),
-
collaboration with the manufacturer to define an optimal risk assessment strategy that meets current regulatory expectations while considering practical constraints (costs, timelines, sample availability),
-
preparation of a comprehensive report outlining the selected strategy.
This service is particularly valuable for manufacturers who struggle with MRI‑related questions; whether about applicable standards, induced risks, or determining worst‑case configurations.
Read the full article on DeviceMed
Healtis’ Commitment to MRI Safety
At Healtis, we support medical device manufacturers throughout their entire MRI evaluation process : from defining the strategy to performing the tests and interpreting the results. Our goal is to ensure a reliable, compliant, and tailored approach for every project.
If you have any questions about this service, reach out at [email protected]!